2018 has been a busy and exciting year for Portal Instruments as we continue to advance our connected, needle-free drug delivery device. As the year draws to a close, we would like to thank our team for their efforts and share with our friends and supporters all that we have accomplished.
Portal continues to advance our connected needle-free injector so that patients can have the option to use an advanced technology for their medication management. Our patented technology has enabled us to build a device that is quiet, fast, and can deliver meaningful volumes of biologic drugs. It is computer controlled for precise delivery, has electronic feedback indicators, and is connected for automatic tracking and reminders.


Our market research and human factor studies continue to support that patients prefer needle-free injections. While pain is the first issue that people think of, there are other challenges with needles that people are looking to avoid, such as the nuisance of sharps disposals, the time it takes to do an injection, and the stigma of having needles in the house. Do you take an injection or know anyone who does? Why do you hate taking shots? Share with us on Facebook at Portal Instruments.

Digital health continues to make great advances in consumer wellness and healthcare. Watches, scales, fitness trackers, CPAPs, asthma inhalers and even thermometers are connected, allowing consumers and patients to track and measure results. Likewise, Portal’s needle-free injector is connected for automatic tracking of injections and reminders. Injection history can be viewed on an app and shared with physicians, which has been reported as a desired feature by patients across categories.1, 2 Portal continues to work with physicians on desired features for our connected injector while ensuring utilization of the latest digital security measures.
News continues to spread about Portal Instruments and the option of a needle-free solution. We have spoken at industry events, such as Partnership Opportunities in Drug Delivery and DeviceTalks; we have been featured in USA Today and won Medical Design and Outsourcing’s Top 10 Hottest Medtech Startups of 2018.
In 2018, we began the transition from an R&D company producing prototypes to a commercial manufacturing company. Our manufacturing team is now ramping up to meet the next level of volume requirements.
Quality is of the utmost importance to us as patients trust us to reliably and safely deliver their medications. With that in mind, we transitioned our Quality System to ISO 13485: 2016.
Outside of work, Portal employees celebrated the end of 2017 at the Annual Boston Christmas Cavalcade for the homeless, helped out at the The Greater Boston Food Bank, participated in the local Crohn’s and Colitis Foundation walk and supported the City Mission Coat Drive.
We’ve done a lot this year!
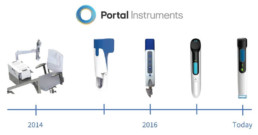
But looking back even further, it was only a little over four years ago that Portal Instruments was spun out of Dr. Ian Hunter’s lab at the Massachusetts Institute of Technology. Since then, Portal’s needle-free drug delivery device has evolved from a bench-top system to a clinical proof of concept, miniaturized to a handheld version and is now on its way to commercialization. We are proud that as a medical device company we have been able to conduct clinical studies in less than 2 years from inception and ramp up for commercial production within 5 years.
There are so many patients out there who are suffering from chronic illnesses and we continually ask ourselves how we can improve the lives of patients who have to take medications regularly. For them, we are all inspired to make a difference in drug delivery and disease management.

Happy Holidays and best wishes for a healthy, happy, and prosperous 2019!
1 King, Diane K., et al. “What patients want: relevant health information technology for diabetes self-management.” Health and Technology 2.3 (2012): 147-157.
2 Wilcox, Lauren, et al. “Interactive tools for inpatient medication tracking: a multi-phase study with cardiothoracic surgery patients.” Journal of the American Medical Informatics Association 23.1 (2016): 144-158.
Pages of the Portal Instruments website may contain links to third party websites that are not under the control of Portal Instruments. Portal Instruments is not responsible for the content of any such site and the inclusion of these links does not imply the endorsement of the content of the linked site by Portal Instruments.
Portal’s platform is in development and not available for sale or use.

